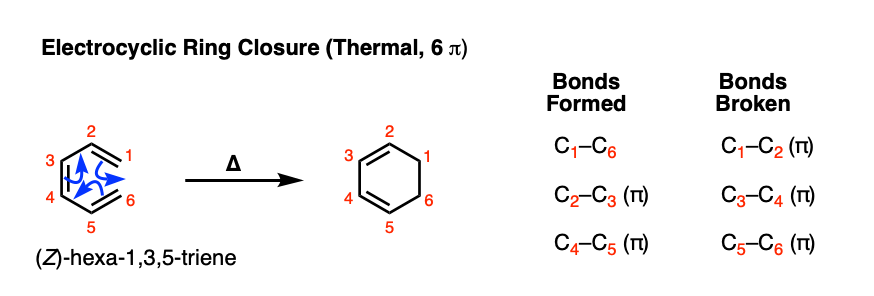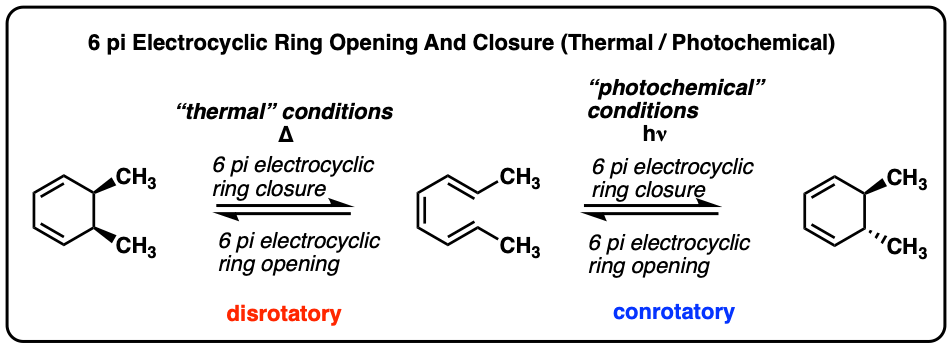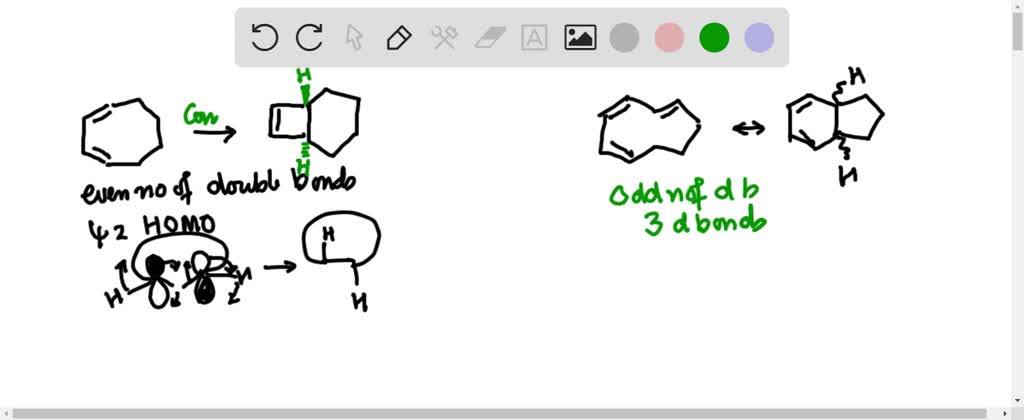
SOLVED:a. Identify the mode of ring closure for each of the following electrocyclic reactions. b. Are the indicated hydrogens cis or trans?
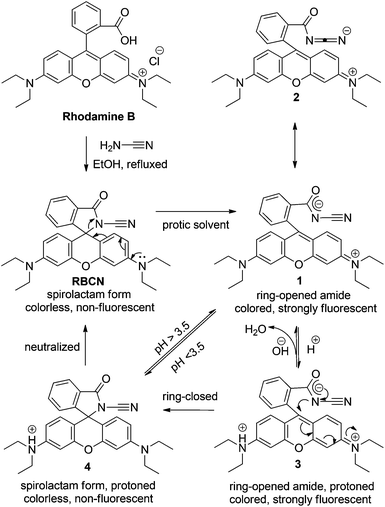
An acid catalyzed reversible ring-opening/ring-closure reaction involving a cyano-rhodamine spirolactam - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C3OB27356C

The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from Cyclodecadiene Q or by photochemical electrocyclic ring closure from Cyclodecadiene R. Draw the structures of Q and R, and

Consider the following electrocyclic ring closure. Does the product form by a conrotatory or disrotatory process? Would this reaction occur under photochemical or thermal conditions? | Homework.Study.com

Michael–Michael Ring-Closure Reactions for a Dihapto-Coordinated Naphthalene Complex of Molybdenum | Organometallics

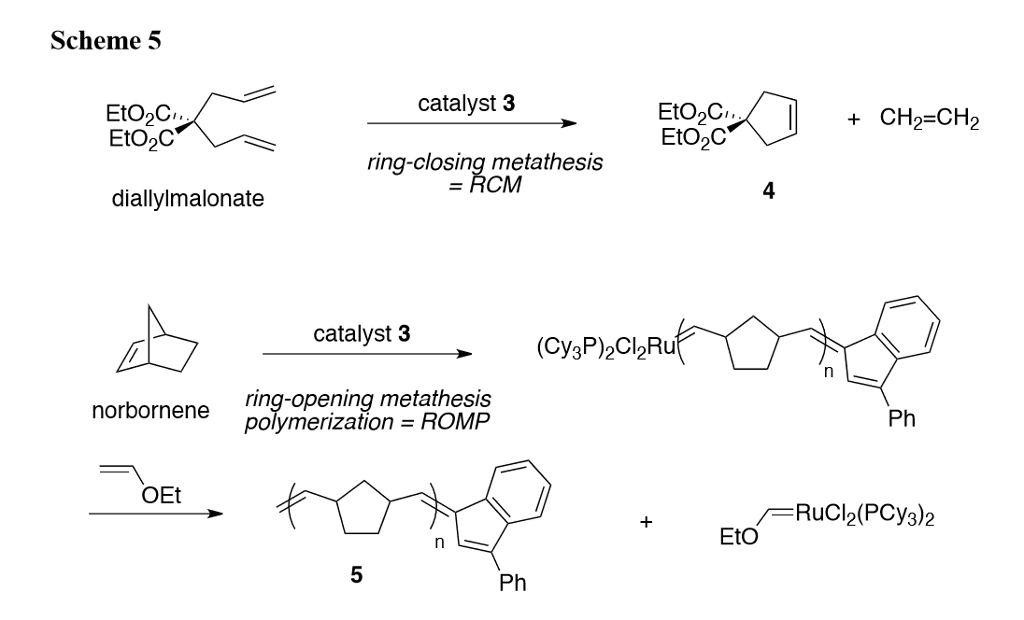
Ring-Closing Metathesis of Allylsilanes/Electrophilic Desilylation To Prepare exo-Methylidenecycloalkanes. Short Syntheses of Teucladiol and Poitediol | Journal of the American Chemical Society

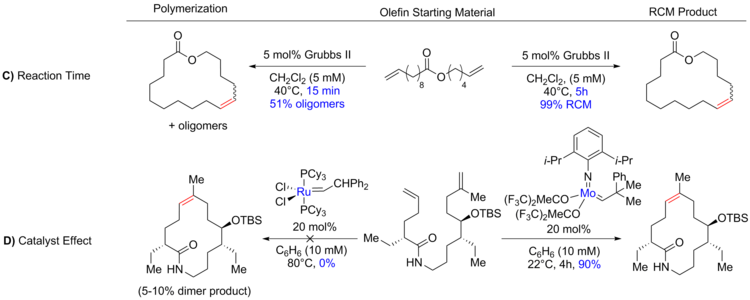
FeCl3‐Catalyzed Ring‐Closing Carbonyl–Olefin Metathesis - Ma - 2016 - Angewandte Chemie International Edition - Wiley Online Library